Elias James Corey Ph.D.
The Nobel Prize in Chemistry 1990
Organic Chemist - Interested in application of organic chemistry to human health. His groups have achieved a multitude of total syntheses of complex molecules. Originator of retrosynthetic analysis. Recognized as "Most Cited Author in Chemistry" by American Chemical Society in 2002.
"Organic chemistry was especially fascinating with its intrinsic beauty and its great relevance to human health."
Biography
by Dan Lednicer PH.D. Organic/Medicinal Chemist
Nobel prizes in many of the sciences are awarded in recognition of some singular achievement. The Nobel Prize in Physics for example may be awarded to a relatively young scientist whose investigations provide a new insight to the nature of matter. The Nobel Prize for Chemistry covers the various specialized areas of this broad scientific discipline. Organic chemistry, that is, the chemistry of carbon compounds, comprises one of those disciplines. This field differs from many of the other sciences by the fact that it comprises of a large body of settled science. It is consequently unlikely that an organic chemist will come up with some singular achievement. The organic chemists who have been awarded a Nobel Prize have instead been selected for a body of many accomplishments achieved during the awardee's career. The Prize has also sometimes recognized the development of new reagents which have made possible hitherto difficult transforms; this usually takes place only after many years after the invention when the method has demonstrated its utility. The 1990 Nobel Prize in Chemistry was consequently awarded to E.J. Corey for "…his important contributions to synthetic organic chemistry".
Elias James Corey was born in Methuen, Massachusetts in 1928. Graduating from high school in 1945 at the tender age of 16, he immediately enrolled in MIT. Although he originally intended to major in engineering, his enthusiasm was roused by a required course in chemistry. In his own words: "Organic chemistry was especially fascinating with intrinsic beauty and great relevance to human health". Corey stayed on at MIT after graduating in 1948 with a BS in chemistry; he had been invited by John Sheehan to join that professor's research group. The group was at that time focused on investigating the chemistry aimed at producing the then-new antibiotic penicillin by chemical synthesis as well as analogues of the compound. The chemical structure of the compound incorporated some previously unknown features that contributed to the fragility of the antibiotic to many chemical reagents. Laboratory experiments aimed at preparing penicillin analogues by chemical synthesis provided Corey a foretaste of the chemistry that would occupy his career. He completed his thesis work for a Ph.D. in just over two years. In 1950 Corey was appointed Instructor in the Chemistry Department of the University of Illinois in Champaign-Urbana. He rose through the ranks, becoming Assistant Professor in 1954 and the full Professor in 1956 at the unusually early age of 27.
The research projects carried out by Corey himself and a group of graduate student and post-doctoral fellows during his first years at Illinois included detailed investigations of the mechanisms of some organic reactions. The menu of his group's projects grew to include determination of the detailed three-dimensional chemical structures of complex molecules isolated from plants. Several projects on the preparation of natural products by total synthesis foreshadowed the future directions of Corey's focus. In 1957 he took Sabbatical leave enabled by a Guggenheim Fellowship, dividing his time between Harvard and Switzerland, England and Sweden. He learned of the then-just emerging field of prostaglandins on his visit to Sune Bergtrom's laboratory in Lund in Sweden. This class of hormone-like compounds would form one his group's major projects in coming years. Just two years later, in 1959 Corey received an offer of a Professorship in the Chemistry Department at Harvard. That department was at that time one of the real hotbeds of organic chemistry hosting a group of distinguished organic chemists. Corey moved to Cambridge in 1960 where he spent the rest of his career at Harvard.
Once at Harvard, Corey's research turned increasingly towards chemical synthesis. The size of his research group grew accordingly accommodating dozens of graduate students and postdoctoral fellows at any given time. He retired from his professorship in 1998 at the mandatory age of 70. Since faculty cannot train graduate students after they retire the research group from then on consisted of only postdoctoral fellows. Over the course of his career his group has completed multistep syntheses of more than 250 target compounds, many of which are natural products. The group has also produced many new chemical reagents and methods for carrying out syntheses. Some of the chemists who got their start in his laboratory went on to become distinguished academic chemists in their own right. The work is recorded in excess of 1000 papers in chemical journals. He also served as co-author on several books on chemical synthesis. Corey has received numerous awards; the most prestigious include U.S National Medal of Science, the Japan Prize in Science, and the Priestley Medal of the American Chemical Society.
Organic synthesis comprises the process of building molecules of organic compounds. This usually involves a step-by-step process in which a molecule is assembled from smaller fragments. Some steps may be devoted to modifying the intermediate to provide a site for attaching a given fragment. Each of those steps comprises a chemical reaction where the intermediate and fragment will be allowed to interact under specified conditions in the presence of reagents or catalysts. The product from a reaction will usually be purified before being subjected to the next step. A synthesis may be as short as a single step or may on the other hand require dozens of discrete steps. The literature of organic chemistry lists a very large set of reactions; this will determine the design of a synthesis because it indicates which transforms can be performed. Syntheses had been traditionally designed in a forward direction. The chemist would build the step-by-step scheme from available starting material to the target molecule using known reactions. Corey introduced the powerful concept of retro-synthesis for designing such routes. This in essence turned the process of designing a synthesis on its head. In retro-synthesis the target molecule is disassembled on paper by breaking the chemical bonds that might have been built in the final step. This of course also draws on the list of known reactions. The process is then repeated; each of the resulting fragments is disassembled by breaking bonds. This process is reiterated until the scheme displays readily available starting materials. Since there may be available more than a single breakable point at any stage the process can create alternate routes. The overall result would be a tree of possible routes to the target compound. In its initial form the value of the process depended on the practitioner's memory for organic reactions. A digital computer version of retro-synthesis has at its command a vast selection of organic reactions and available starting compounds for carrying out the necessary disconnections. Wikipedia currently lists eight Computer-assisted organic synthesis (CAOS) programs.
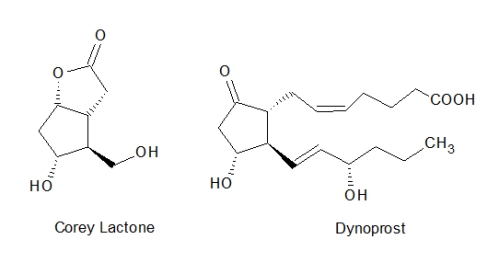
A major program in Corey's group has involved the prostaglandins and the related eicosanoids. The former comprise a group of hormone-like compounds involved in many inflammatory processes. Several synthetic analogues have been approved as therapeutic drugs. The naturally occurring prostaglandin-E2, also known as dynorostone is used in inducing labor. The modified prostaglandin analogue mifepristone was initially approved for avoiding the stomach irritation from non-steroid anti-inflammatory drugs. It has more recently been used for inducing abortions in conjunction with mifepristone (RU 486). The synthetic version of a naturally occurring prostaglandin, alphaprostol is approved for treating erectile dysfunction. A key intermediate developed in Corey's laboratory for synthesizing prostaglandins, widely known as the "Corey lactone" has been adopted by other labs involved in the synthesis of prostaglandin analogues.
Even the best planned syntheses will often meet difficulties where a planned step goes wrong. It is necessary at such an impasse to develop either a new reagent or even a new reaction that will solve the problem. Corey's group has been especially productive in devising new methodology. Organic chemists pay inventors of new reagents or reactions a signal honor when they name after them the new methodology. The literature accordingly lists the "Corey-Suggs Reagent" (pyrdinium chlorochromate for oxidation), the Corey-Chaykovsky reagent (timehylsulfoxonium iodide for adding a carbon atom), the Corey-oxazaborolidine (for stereoselective reduction). As for new methodology there is the Corey-Seebach reaction, the Corey-Chaykovsky reaction and the Corey-House reaction.
Impossible Dreams by EJ Corey, J. Org. Chem, Vol. 69, No. 9, 2004, 2919
 ABOVE AND BEYOND ORGANIC SYNTHESIS, Elias J. Corey personifies extraordinary intellectual acumen and traditional values of family, service, and looking out for others by A. Maureen Rouhi, Chemical and Engineering News, Washington
ABOVE AND BEYOND ORGANIC SYNTHESIS, Elias J. Corey personifies extraordinary intellectual acumen and traditional values of family, service, and looking out for others by A. Maureen Rouhi, Chemical and Engineering News, Washington

Discover Your Abilities and Aspirations!
 $10 $25 $50 $100 Other
$10 $25 $50 $100 Other
Tax Exempt 501(c)3 Non-Profit Organization
Any Currency
“…the peace that is found in libraries and laboratories…” - Louis Pasteur
Copyright © 2023 Ganga Library Inc. All Rights reserved.;

Photo: Elias .J. Corey at Harvard. Creator: Trvthchem personal photo. 19 Nov 2007. Source: Wikimedia Commons.
Name: Elias James Corey
Birth: 12 July 1928, Methuen, MA, USA
Institution: Harvard University, Cambridge, MA, USA
Award: "for his development of the theory and methodology of organic synthesis"
Subject: organic chemistry
National Medal of Science Chemistry 1988
Biography
Nobel Lecture
Publications
Books
Patents
Thesis
History of Discovery
Honoring Corey
Images
Videos













